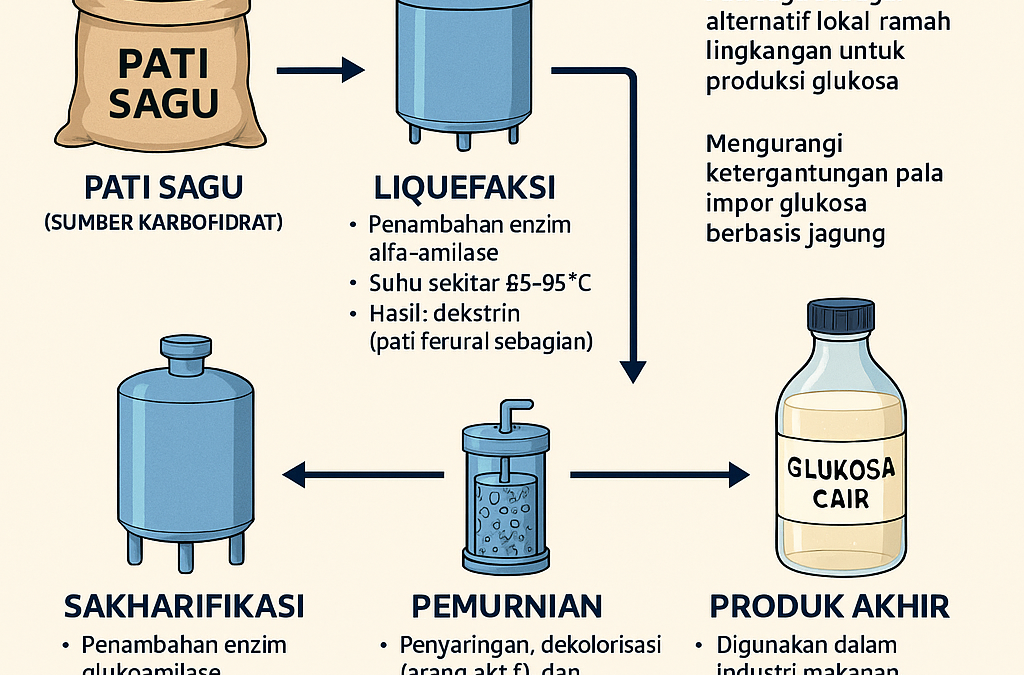
Indonesia is the world’s largest producer of sago (Metroxylon sp.). Indonesia’s sago land area reaches 51% of the world’s sago land area, which is 1.25 million hectares. Sago palms are the highest producers of dry starch compared to rice and corn. Sago can produce 25 tons of dry sago per hectare, while rice and corn produce 6 and 5.5 tons per hectare, respectively. Another advantage of the sago plant compared to other starch-producing plants is that sago cultivation technology is relatively easy, simple, and more economical. As the largest starch producer, sago has great potential to be developed as a food source and raw material for synthesizing starch-derived products. One of the derivative products of sago starch is liquid glucose, as developed thru an innovative idea by the Department of Industrial Chemical Engineering Technology (TRKI) Vocational Undip, namely Mohamad Endy Julianto, Malika Pintanada Kaladinanty, and Abitha Mona Wisya.

Malika stated that liquid glucose is currently made from cassava and corn. Liquid glucose is used as a substitute for granulated sugar in the confectionery, beverage, and biscuit industries. Liquid glucose can improve smoothness, lower the freezing point, prevent microbial spoilage, and improve the texture of ingredients. Domestic demand for liquid glucose is only about 60% met. Indonesia’s glucose syrup imports reached 112,396 kg. The industrial world’s need for sweeteners continues to increase along with the development of the industries that use them, Malika explained.
To synergize efforts in diversifying and developing sago-based products and in increasing national self-reliance in meeting sweetener needs, especially liquid glucose, it is necessary to develop a process for producing liquid glucose from sago starch, said Abitha. Abitha revealed that liquid glucose is made thru the process of starch hydrolysis. The hydrolysis process can occur thru acid or enzymatic hydrolysis. In the production of glucose syrup, the selection of the starch source must consider its amylose and amylopectin content. A starch source with high amylopectin is better because it has insoluble starch particles (ISPs) that can be hydrolyzed both acidically and enzymatically. Starch must also be low in protein and fat because it causes the Maillard reaction, which can cause a brownish color in liquid glucose. Sago starch has a fairly high amylopectin content and is low in protein and fat, making it suitable as a raw material for producing liquid glucose, said Abitha.
Meanwhile, Endy added that enzymatic hydrolysis of sago starch in a tubular membrane reactor appears very promising for development. Enzymatic hydrolysis has many advantages compared to conventional hydrolysis processes using acid. These advantages include higher conversion of glucose to D-glucose (which can reach 97%), lower operating temperatures, no need for corrosion-resistant equipment, enzymatic hydrolysis does not affect product color, it is economical in terms of chemicals because it does not require neutralizing chemicals, and no salt deposits are produced.

Endy stated that membrane reactors combine two processes: enzymatic saccharification and separation. The membrane separation process will provide selectivity for separating large particles based on their physical dimensions. During the hydrolysis process, the hydrolysis products will be separated with simultaneous recycling of undigested substrate and enzyme. Enzymes can be discovered and reused. One of the main advantages of using membrane reactors is the simultaneous separation and removal of inhibitor compounds from the reaction medium thru the membrane pores. The substrate is continuously fed into the reactor to compensate for the permeate flux and maintain a constant reactor volume. The application of membrane reactor in the hydrolysis process allows for modification of the reaction conditions, including pH, temperature, starch concentration, enzyme dosage, and Endy cap.